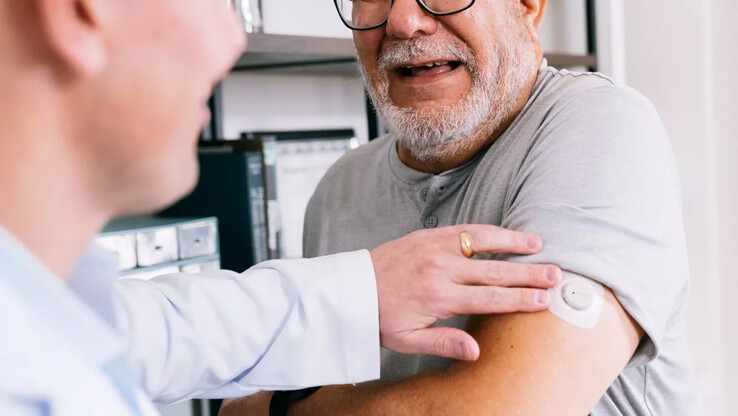
The Dexcom Stelo has become the first continuous glucose monitor (CGM) of its kind to receive FDA clearance in the US. It is an over-the-counter (OTC) solution, meaning it will be available without a prescription. According to Dexcom, its new product will help certain groups of people track their blood sugar levels and the impact of lifestyle factors.
The Dexcom Stelo Glucose Biosensor System allows you to track your blood glucose levels in the accompanying app, with measurements taken every 15 minutes. The sensors are applied to the back of the arm with a patch and must be changed every 15 days. Personalized insights in the smartphone app will help users discover trends in their blood sugar levels and how they are affected by diet and exercise. Presumably, data collected by the Stelo will also be viewable on smartwatches like the Garmin Forerunner 265 via the Connect IQ Dexcom app.
The FDA notes that the solution is designed for people over 18 without diabetes or individuals treating Type 2 diabetes with oral medication. It is not suitable for those treating diabetes with insulin and should not be used in place of a medical professional. The Dexcom Stelo CGM will launch in the US this summer, though the pricing has yet to be announced. As an idea, a bundle of three Dexcom G7 Sensors retails for $377.73 at Amazon Pharmacy.