It appears PKvitality, a French company that is based in Paris, has made excellent progress with its K’Watch Glucose CGM smartwatch, which has been developed to provide a blood-free and painless option for taking regular blood sugar level measurements for those having to manage their lives with diabetes. The startup took the K’Watch Glucose device into its first-in-human clinical trials (third trial overall) in November 2021, with the test being carried out at the AMCR Institute in California. The third trial results were published as part of a press release not too long ago (July 26), and they are good enough to have diabetics at least feeling cautiously optimistic.
The device utilizes an “array of micropoints and biosensors” that can take blood glucose measurements without causing any pain. Apparently, the K’Watch Glucose CGM smartwatch produced a 29% margin of error on accuracy (MARD: Mean Absolute Relative Difference) on its first run, and the developers were able to reduce this to 19% for the second trial. The end result for the third trial was a MARD level of 16%, with changes in the measuring algorithm and K’apsul patch refinements helping the K’Watch reach this promising level of accuracy. PKvitality’s founder, Luc Pierart, was particularly effusive about the clinical trial’s results:
This tremendous acceleration towards the precision required in our clinical trials makes us confident in the development of our product. We are a few months away from validating our project and offering a CGM that meets the expectations of diabetes patients: painless, without adhesive issues, discreet and ultra-practical.
Obviously, the things that the millions of people out there with diabetes who are tired of using lancets and testing strips want to know is when, how much, and will it be covered by insurance. It seems PKvitality has “around 2024” in mind for a K’Watch Glucose launch, and the price of the device is currently expected to be $199 with the replaceable K’apsul Glucose sensors costing about $99.90 a month. Prices are, of course, subject to change and regional variations. Unfortunately, there is no clear-cut answer in regard to insurance as it is still early days, with the company’s FAQs suggesting it will depend on variables such as reimbursement coming from local agencies and providers.
Source(s)
PKvitality (1/2/3) & trial results (pdf)
All images and video: PKvitality
Paris, July 26, 2022 - The French startup PKvitality, focusing on health and sport
biowearables, is developing K'Watch Glucose, the first Continuous Glucose Monitor (CGM)
in the form of a smartwatch, designed to change the daily lives of millions of diabetic
patients. After two first phases of testing which brought a significant step forward on the
initial roadmap, the company champion of innovation against diabetes announces the
results of its third phase of clinical trials on humans (First-in-human).
PKvitality's mission is to enable everyone to contribute actively to their well-being and health
through products that are simple and comfortable to use. Thus, its K'Watch Glucose
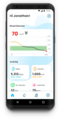
smartwatch will be the world's first CGM device that will track glucose levels discreetly and
continuously at the wrist. This system, currently at the stage of clinical studies, will measure
the level of glucose in the interstitial fluid through micro-tips. It will thus offer a painless and
non-stigmatizing experience to the user.
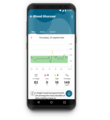
The 3rd clinical trial on humans that announces the success of K'Watch
After two conclusive clinical trials, the results of the 3rd study suggest a promising future for
the development of K’Watch. Indeed, the measurement of glucose levels for patients with
type 1 and type 2 diabetes is commonly a demanding process whether in terms of accuracy,
tolerance, or pain. Based on these criteria, the objective has been more than achieved since
K’Watch obtains a 16% margin of error on the accuracy (MARD) for continuous glucose
measurement against the 17-18% initially desired. This efficiency is the result of in-depth
improvements in the patch and the algorithm.
In the past few months, PKvitality has also unveiled the results of its 1st and 2nd clinical studies
on humans. The results of these studies were a real success and showed 19% accuracy in
continuous glucose analysis. In addition, studies conducted by PKvitality reveal an excellent
user experience with very good tolerance for patients with no adverse effects, pain, or
irritation during use.
The trials took place at the AMCR Institute, a clinical research center focused on diabetes and
obesity with worldwide renowned expertise in medical devices related to metabolism.
According to Luc Pierart, founder of PKvitality, "With
K'Watch, we seek to offer people with diabetes a
technological innovation that will accompany them in their
daily lives. Although we started clinical trials at the end of
2021, we are making rapid and very conclusive progress.
This tremendous acceleration towards the precision
required in our clinical trials makes us confident in the
development of our product. We are a few months away
from validating our project and offering a CGM that meets
the expectations of diabetes patients: painless, without
adhesive issues, discreet and ultra-practical.”
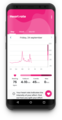
K’Watch for diabetics
PKvitality develops K'Watch, the world's first Continuous
Glucose Monitor (CGM) in a form of a smartwatch, able to
measure and display glucose levels continuously and
discreetly, for patients with diabetes. His system measures glucose levels by analyzing
interstitial fluid through micro-tips. It provides a painless, non-stigmatizing experience and
solves the adhesive problem. In addition, PKvitality has also joined the largest e-health
accelerator in Europe, Future4Care.
About PKvitality
PKvitality is a health & sport bio-wearable start-up currently working on the K’Watch Glucose,
a Continuous Glucose Monitoring (CGM) device in the form of a smartwatch currently in
clinical trials. It will enable a painless, discreet and precise monitoring of systemic glucose level
anytime and anywhere. Using the same technology, PKvitality is also working on K’Watch
Athlete, a smartwatch which will provide real-time monitoring of lactic acid – an indicator of
muscle fatigue – to significantly improve training and performance among athletes.