The KnowU and UBand Bio-RFID non-invasive blood glucose monitors are currently under further testing by the company that designed them, Know Labs (Visulant, Incorporated). While the two devices are still in a pre-FDA study stage (live “scientific research validation”), recent comparison tests published by the company have shined a very promising light on the KnowU and UBand. Both monitors utilize radio-frequency identification (RFID) to collect the required data for blood sugar level management.
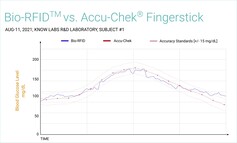
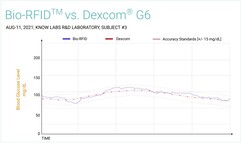
Charts produced by Know Labs (see below) compare its Bio-RFID technology with the Accu-Check Fingerstick, FreeStyle Libre 14-day, and Dexcom G6, which all rely on minimally invasive/invasive methods to produce blood glucose level readings. While Bio-RFID did cross the margins for accuracy standards briefly against the Accu-Check Fingerstick, the overall readings show that the KnowU and UBand are clearly getting closer to being ready for the required clinical trials they need to pass before they can go on the market.
As well as offering a pain-free alternative to finger sticking with lancets, the company has estimated a <US$1,000 per year cost for using a Bio-RFID technology product, which is considerably less expensive than many other solutions that diabetes sufferers have to rely on. The KnowU monitor, which can easily be carried around in a pocket, and the UBand wearable offer users either on-demand or continuous measurement solutions, respectively. Radio waves from the Bio-RFID devices penetrate the user’s tissue and “capture molecular signatures”, meaning the blood glucose monitors can be used by anyone, diabetic or not, who wants to observe their blood sugar levels.
There has been mention of the KnowU and UBand potentially progressing onto the FDA approval stage at some point in 2022, which means painful finger-sticking could be consigned to history’s bin within a couple of years, as the FDA approval process for medical devices is much quicker than it is for drugs, especially for non-invasive products. An accompanying app will be made available for Android and Apple smartphones that will assist users with checking their KnowU or UBand readings, whether it’s just on-demand or continuous blood glucose level management that is required.
Source(s)
Know Labs (& test report pdf) & FierceBiotech & Qualio