Researchers from the Chinese Academy of Sciences have managed to overcome the obstacles that traditional 3D-printing methods present before creating faux cardiac tissue by retrofitting a robotic arm to do the bioprinting. Not only was the novel 3D-printing method successful in fabricating a viable vascular organoid, but the faux heart tissue kept beating for six months after being printed. Called "A multi-axis robot-based bioprinting system supporting natural cell function preservation and cardiac tissue fabrication," the paper lists several advantages of this method in the abstract:
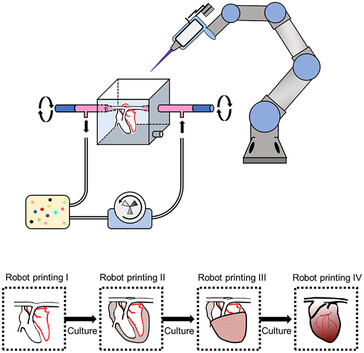
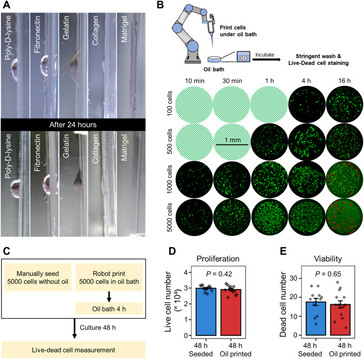
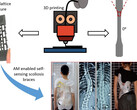
Despite the recent advances in artificial tissue and organ engineering, how to generate large size viable and functional complex organs still remains as a grand challenge for regenerative medicine. Three-dimensional bioprinting has demonstrated its advantages as one of the major methods in fabricating simple tissues, yet it still faces difficulties to generate vasculatures and preserve cell functions in complex organ production. Here, we overcome the limitations of conventional bioprinting systems by converting a six degree-of-freedom robotic arm into a bioprinter, therefore enables cell printing on 3D complex-shaped vascular scaffolds from all directions. We also developed an oil bath-based cell printing method to better preserve cell natural functions after printing. Together with a self-designed bioreactor and a repeated print-and-culture strategy, our bioprinting system is capable to generate vascularized, contractible, and long-term survived cardiac tissues. Such bioprinting strategy mimics the in vivo organ development process and presents a promising solution for in vitro fabrication of complex organs.
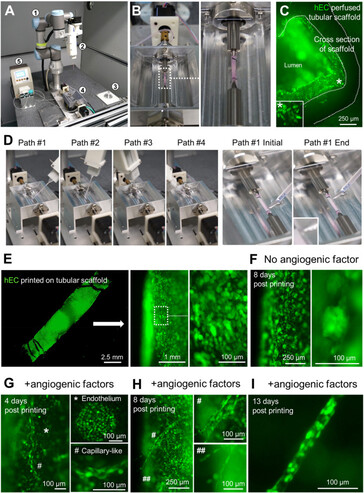
The typical three-dimensional layering print methods aren't suitable for creating complex vascular networks as the stacking often damages the cells while the biomaterials that glue them together prevent the functioning as organ tissue. This is why the researchers programmed a robotic arm with six rotating joints to inject cells with little damage on a scaffold from all sides, creating faux blood vessels down to newly formed capillaries. Not only that, but using two such robotic arms to deposit different cell types such as cardiomyocytes on the scaffold simultaneously, they managed to bioprint "vascularized and contractable cardiac tissues, which maintained alive and beating for over 6 months."
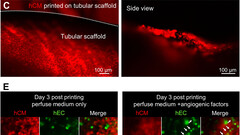
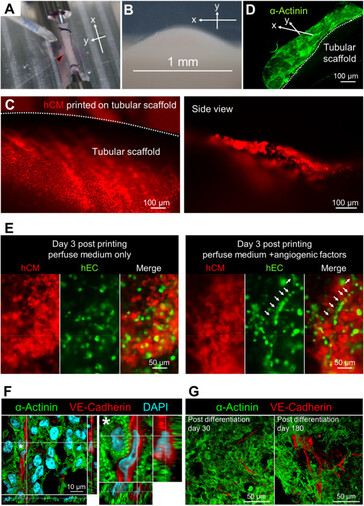
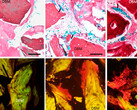
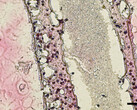
By applying this print-and-differentiation strategy, we fabricated a piece of vascularized cardiac tissue (2 cm in length, 200–500 μm in thickness, and ∼1.256 cm2 total area) on the tubular scaffold, which maintained contraction for at least 6 months. We carried out histological analyses of cardiac tissue on day 30 and day 180 PD, and found no noticeable tissue damage at both time points. In addition, cardiomyocytes were striated with well-organized Z-lines, suggesting that the fabricated cardiac tissue had developed and maintained intact myofibril structures, which is the physiological basic for heart contraction.